Disclaimer: This page contains content specific to Florida Stem Cell Law, which allows specific licensed physicians to administer stem cell therapies that the U.S. Food and Drug Administration has not approved. The law and the content apply to providers licensed in Florida under Chapter 458 (Medical Doctors) and Chapter 459 (Osteopathic Physicians) acting in the course and scope of their employment.
The highest quality, most efficacious, compliant mesenchymal stem cells available.
Wharton’s Jelly mesenchymal stem cells (WJ-MSCs) are multipotent stromal cells isolated from the gelatinous substance within the human umbilical cord known as Wharton’s jelly. These cells are notable for:
Overall, Wharton’s Jelly MSCs represent an excellent source of regenerative cells with applications in tissue engineering, immunotherapy, and regenerative medicine.
In the context of stem cells, “secretome” refers to the complete set of molecules (such as proteins, cytokines, growth factors, exosomes, and other substances) secreted by stem cells into their environment. These molecules are responsible for many beneficial effects of stem cell therapies, including promoting tissue repair, reducing inflammation, and signaling other cells to function or regenerate.
When someone mentions “20 x the secretome,” it typically means that the stem cell product or treatment contains a concentration of secretome components 20 times higher than a baseline, standard, or normal level. This could mean the secretome has been concentrated to be much more potent or is derived from a larger initial number of stem cells, resulting in a higher quantity of the bioactive factors responsible for therapeutic effects.
In summary, “20 x the secretome” indicates a highly concentrated stem cell secretome product that may be intended to have enhanced therapeutic impact compared to a standard secretome preparation.

In the context of stem cell culture, “doubling no more than 12 times” means that the population of stem cells has undergone no more than 12 rounds of doubling since their initial isolation and culture. Each population doubling represents one complete doubling of the cell number (e.g., from 1,000 to 2,000 cells is one doubling). The population doubling level (PDL) is tracked to measure a cell population’s cellular age or total replication history in vitro.
Tracking and limiting PDL is vital because:
In summary, a PDL limit 12 ensures you’re working with stem cells that have not been excessively expanded, helping maintain desirable biological properties and compliance with best practices in research and clinical settings.
When dealing with stem cells, the number of passages means that the cell culture has been split and subcultured four times since the initial plating. Each passage is a process where cells are detached (usually with enzymes or mechanical means) and transferred to new culture vessels with fresh growth media. This helps prevent overcrowding and keeps the cells proliferating in optimal conditions. The passage number counts how many times this transfer process has happened to that cell population.
For instance, stem cells at passage four have been through four subculturing rounds since they were initially isolated or thawed from a stock. The passage number is significant because stem cell characteristics—such as their growth rate, differentiation capacity, and phenotype—can change over successive passages and affect efficacy and potency.
Culturing stem cells on human media—meaning media that do not contain animal-derived components (like fetal bovine serum) but instead use human-derived or fully defined and Xeno-free ingredients—offers several significant advantages:
Overall, human media (or Xeno-free, chemically defined media) provides safety, consistency, and suitability for clinical translation—critical advantages for stem cell research and therapy.
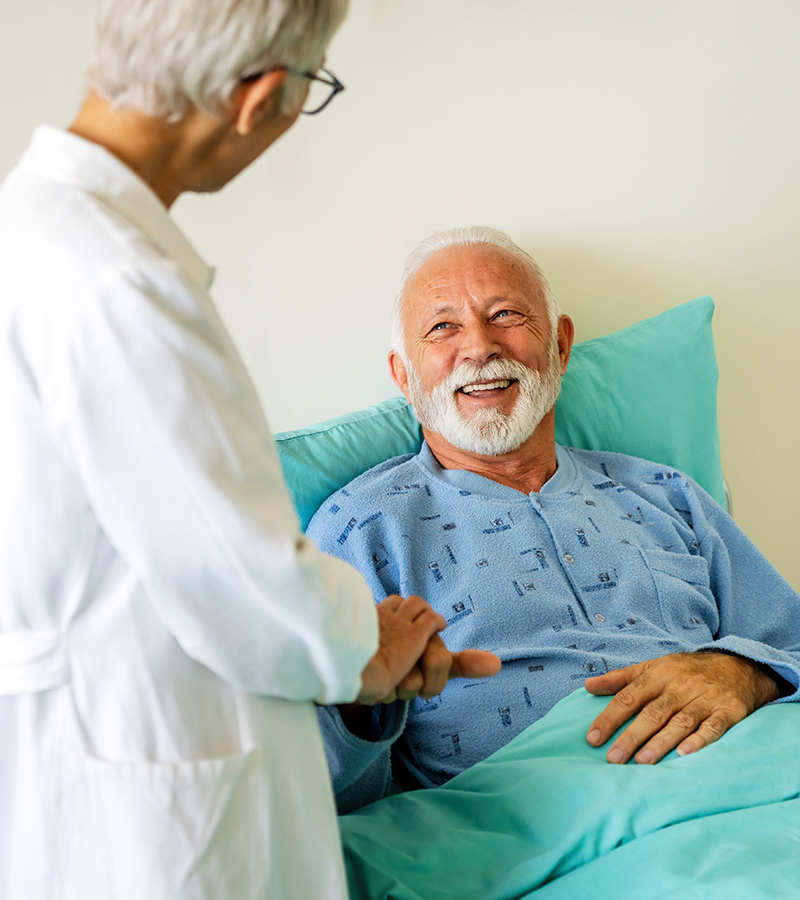
We are committed to delivering exceptional, patient-centered care with compassion, professionalism, and integrity. Our goal is to ensure every patient receives the highest standard of medical attention, using advanced technology and a team of experienced healthcare professionals. Trust us to be your partner in lifelong wellness.
Our skilled medical professionals bring years of expertise to ensure accurate diagnoses and compassionate care.
From routine checkups to specialized treatments, we offer a full range of healthcare solutions under one roof.
Our modern clinics are equipped with the latest medical technology to support advanced and effective care.
With multiple locations and flexible hours, we make it easy for you to access quality healthcare when you need it.









Copyright 2025 © All Right Reserved by MD Clinix